
The CAPA procedure must also assign responsibilities for the various steps. The procedure should detail what information is required to initiate, classify (Critical, Major, Minor), investigate, risk assess, justify, communicate, implement, approve, close, trend, review and document the CAPA. The CAPA procedure must include a detailed description of the information required as there are several steps to take and carefully document before a CAPA can be closed. The importance of a good CAPA procedure and training 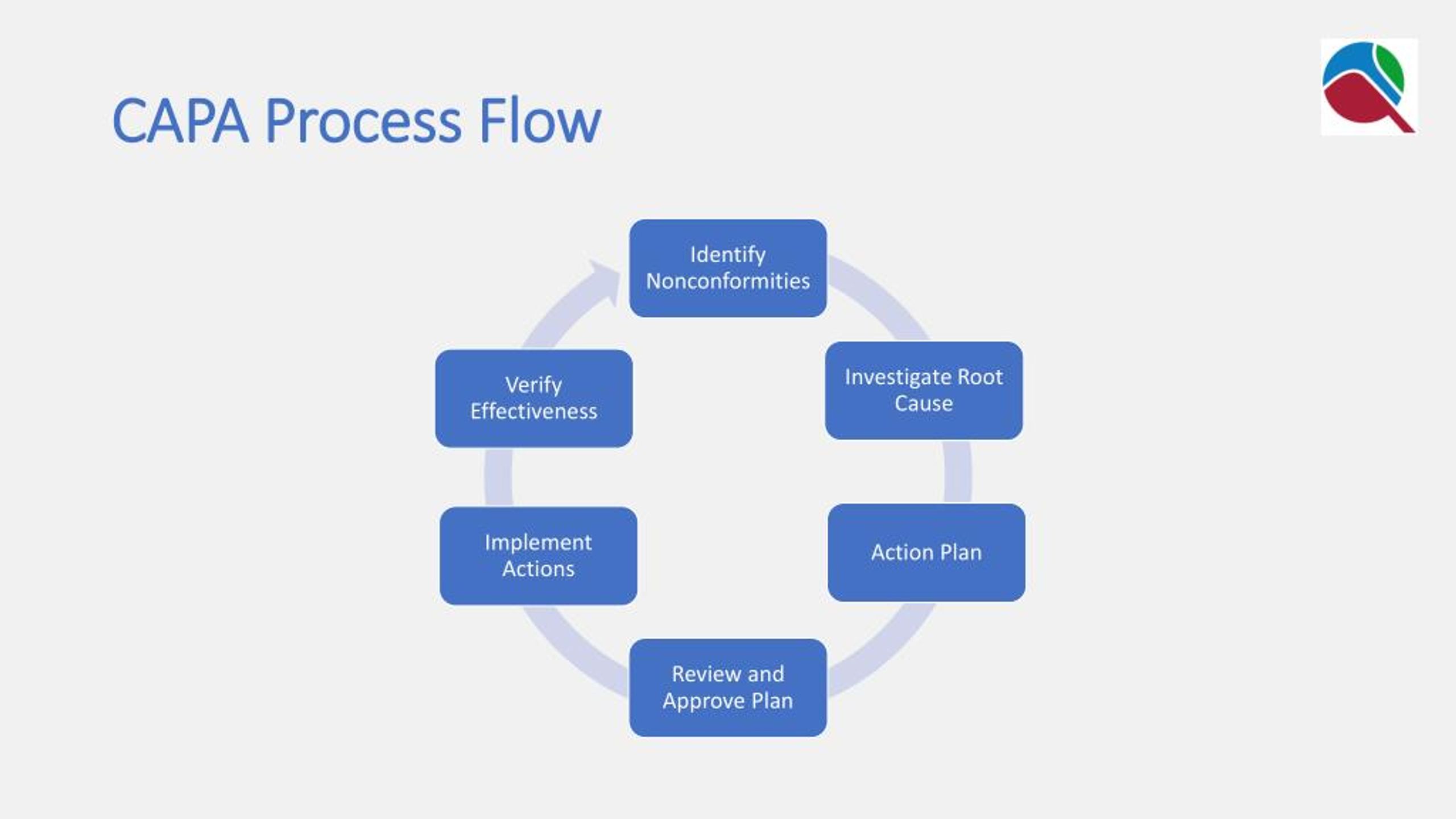
The FDA has proceeded to warn companies that their failure to comply may result in legal action including, but not limited to, seizure, injunction, and civil money penalties.Ī company could go out of business if they do not have a good CAPA procedure and do not prioritise closing CAPAs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
